Activated charcoal is used in water filters, medicines that selectively remove toxins, and chemical purification processes. In the production of steam-activated charcoal, first the coconut shell or coal is heated to create a char. This char is then “activated” in a furnace at high temperatures of 1,700° to 1,800°F with steam in the absence of oxygen. In the steam-activation process, all volatile compounds are removed, and at the same time layer after layer of carbon atoms are peeled off, enlarging the existing internal pores, and leaving behind a carbon skeleton. The carbon + steam reaction results in producing hydrogen gas and carbon monoxide (C+H2O=H2 +CO). As the carbon monoxide gases off it takes carbon atoms with it. Typically, 3 pounds of raw charcoal will produce 1 pound of activated charcoal.
Chemical-activation produces the same end result – a much increased internal surface area – but uses a chemical solution to chew away the internal structure. Typically, wood-based activated charcoals are commercially made using heat (450–900 °C) and phosphoric acid. This “activation” process results in the creation of an enormous surface area — on the order of 600 to 1,200 square meters per gram (m2/g), depending upon the raw material used and the process.
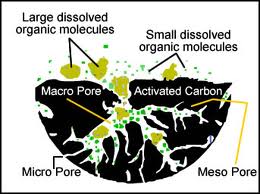
The treatment results in a highly porous charcoal. These tiny holes give the charcoal a surface area of 300-2,000 m2/g, allowing liquids or gases to pass through the charcoal and interact with the exposed carbon. The carbon adsorbs a wide range of impurities and contaminants, including chlorine, odours, and pigments. Other substances, like sodium, fluoride, and nitrates, are not as attracted to the carbon and are not filtered out. Because adsorption works by chemically binding the impurities to the carbon, the active sites in the charcoal eventually become filled. Activated charcoal filters become less effective with use and have to be recharged or replaced. Organics are typically removed by adsorption and residual disinfectants by catalytic reduction.
Several factors influence the effectiveness of activated charcoal. The pore size and distribution vary depending on the source of the carbon and the manufacturing process. Large organic molecules are absorbed better than smaller ones. Adsorption tends to increase as pH and temperature decrease. Contaminants are also removed more effectively if they are in contact with the activated charcoal for a longer time, so flow rate through the charcoal affects filtration. The quality and use of activated carbon is evaluated on a number of criteria dependent on their intended use. Here are a few of the most common properties important for manufacturers.